Troponin I (cardiac) Antibody
Rabbit polyclonal antibody
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB |
|---|---|
| Primary Accession | P48787 |
| Reactivity | Mouse, Rat |
| Host | Rabbit |
| Clonality | polyclonal |
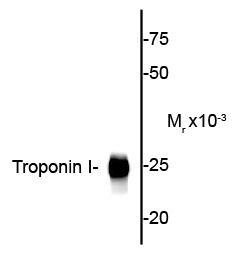
| Calculated MW | 25 KDa |
| Gene ID | 21954 |
|---|---|
| Gene Name | TNNI3 |
| Other Names | Troponin I, cardiac muscle, Cardiac troponin I, Tnni3 |
| Target/Specificity | Fusion protein of complete mouse cardiac troponin 1. |
| Dilution | WB~~ 1:2000 |
| Format | Unpurified neat serum. |
| Antibody Specificity | Specific for the ~25k cardiac troponin I protein. |
| Storage | Maintain refrigerated at 2-8°C for up to 6 months. For long term storage store at -20°C in small aliquots to prevent freeze-thaw cycles. |
| Precautions | Troponin I (cardiac) Antibody is for research use only and not for use in diagnostic or therapeutic procedures. |
| Shipping | Blue Ice |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
Troponin I (cTnI) is 1 of 3 subunits, along with troponin C (TnC) and troponin T (TnT) of troponin complex found in cardiac muscle. cTnI binds to actin in thin myofilaments to hold the troponin-tropomyosin complex in place. Phosphorylation of cardiac isoform of TnI at serines 22,23 in the unique amino-terminal end molecule decreases the calcium sensitivity of the sarcomere, promotes calcium dissociation from troponin C and by extension enhances rates of cross-bridge cycling and diastolic relaxation (Noland, Jr. et al., 1995; Noland et al., 1989). In addition, studies using reconstituted fibers and mutational analysis have shown that PKC phosphorylation of TnI (largely at Ser43) inhibits the actin-cross bridge reaction and reduces the Ca++ dependent actomyosin ATPase rate as well as the calcium sensitivity of force generation (Noland, Jr. and Kuo, 1991). Phosphorylation at Thr144 (mediated by several PKC isoforms) reduces maximal tension development and cross-bridge cycling rates (Sumandea et al., 2008). Importantly, changes in the phosphorylation at each of these sites have been shown to be stage-specific with regard to cardiac disease progression (Walker et al., 2010).
References
Noland TA, Jr., Guo XD, Raynor RL, Jideama NM, Averyhart-Fullard V, Solaro RJ, Kuo JF (1995) Cardiac troponin I mutants - Phosphorylation by protein kinases C and A and regulation of Ca2+-stimulated MgATPase of reconstituted actomyosin S-1. J Biol Chem 270: 25445-25454.
Noland TA, Jr., Kuo JF (1991) Protein kinase C phosphorylation of cardiac troponin I or troponin T inhibits Ca2(+)-stimulated actomyosin MgATPase activity. J Biol Chem 266: 4974-4978.
Noland TAJr, Raynor RL, Kuo JF (1989) Identification of sites phosphorylated in bovine cardiac troponin I and troponin T by protein kinase C and comparative substrate activity of synthetic peptides containing the phosphorylation sites. J Biol Chem 264: 20778-20785.
Sumandea MP, Rybin VO, Hinken AC, Wang C, Kobayashi T, Harleton E, Sievert G, Balke CW, Feinmark SJ, Solaro RJ, Steinberg SF (2008) Tyrosine phosphorylation modifies protein kinase C delta-dependent phosphorylation of cardiac troponin I. J Biol Chem 283: 22680-22689.
Walker LA, Walker JS, Ambler SK, Buttrick PM (2010) Stage-specific changes in myofilament protein phosphorylation following myocardial infarction in mice. J Mol Cell Cardiol 48: 1180-1186.
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.





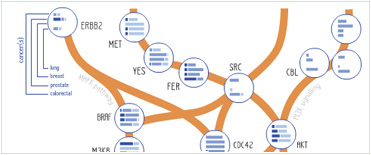







 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.