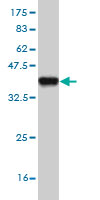
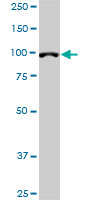
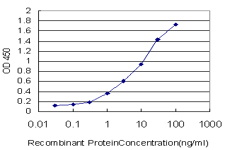
DPYD Antibody (monoclonal) (M01)
Mouse monoclonal antibody raised against a partial recombinant DPYD.
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB, IF, E |
|---|---|
| Primary Accession | Q12882 |
| Other Accession | NM_000110 |
| Reactivity | Human |
| Host | mouse |
| Clonality | Monoclonal |
| Isotype | IgG1 Kappa |
| Clone Names | 7D4 |
| Calculated MW | 111401 Da |
| Gene ID | 1806 |
|---|---|
| Other Names | Dihydropyrimidine dehydrogenase [NADP(+)], DHPDHase, DPD, Dihydrothymine dehydrogenase, Dihydrouracil dehydrogenase, DPYD |
| Target/Specificity | DPYD (NP_000101, 1 a.a. ~ 110 a.a) partial recombinant protein with GST tag. MW of the GST tag alone is 26 KDa. |
| Dilution | WB~~1:500~1000 IF~~1:50~200 E~~N/A |
| Format | Clear, colorless solution in phosphate buffered saline, pH 7.2 . |
| Storage | Store at -20°C or lower. Aliquot to avoid repeated freezing and thawing. |
| Precautions | DPYD Antibody (monoclonal) (M01) is for research use only and not for use in diagnostic or therapeutic procedures. |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
The protein encoded by this gene is a pyrimidine catabolic enzyme and the initial and rate-limiting factor in the pathway of uracil and thymidine catabolism. Mutations in this gene result in dihydropyrimidine dehydrogenase deficiency, an error in pyrimidine metabolism associated with thymine-uraciluria and an increased risk of toxicity in cancer patients receiving 5-fluorouracil chemotherapy. Two transcript variants encoding different isoforms have been found for this gene.
References
Variants in the dihydropyrimidine dehydrogenase, methylenetetrahydrofolate reductase and thymidylate synthase genes predict early toxicity of 5-fluorouracil in colorectal cancer patients. Kristensen MH, et al. J Int Med Res, 2010 May-Jun. PMID 20819423.Genetic polymorphisms associated with 5-Fluorouracil-induced neurotoxicity. Kim SR, et al. Chemotherapy, 2010. PMID 20714149.Value of gene polymorphisms as markers of 5-FU therapy response in stage III colon carcinoma: a pilot study. Fari?a-Sarasqueta A, et al. Cancer Chemother Pharmacol, 2010 Jul 28. PMID 20665215.Absence of large intragenic rearrangements in the DPYD gene in a large cohort of colorectal cancer patients treated with 5-FU-based chemotherapy. Par? L, et al. Br J Clin Pharmacol, 2010 Aug. PMID 20653680.[Correlation between clinicopathological factors and enzymatic activity of orotate phosphoribosyl transferase (OPRT), dihydropyrimidine dehydrogenase (DPD) in esophageal cancer] Takemura M, et al. Gan To Kagaku Ryoho, 2010 Jul. PMID 20647710.
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.











 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.