Anti-Emerin Picoband Antibody
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB, IHC-P |
|---|---|
| Primary Accession | P50402 |
| Host | Rabbit |
| Reactivity | Human, Mouse, Rat |
| Clonality | Polyclonal |
| Format | Lyophilized |
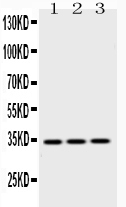
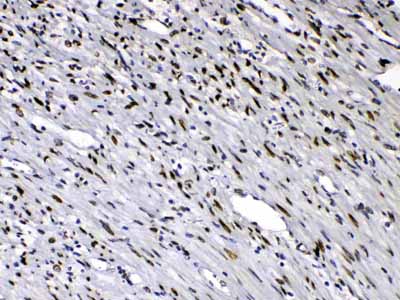
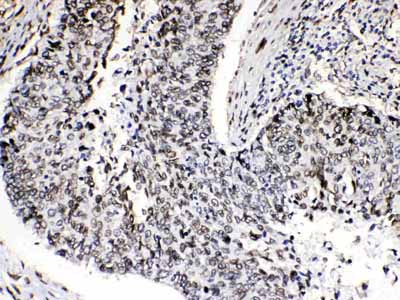
| Description | Rabbit IgG polyclonal antibody for Emerin(EMD) detection. Tested with WB, IHC-P in Human;Mouse;Rat. |
| Reconstitution | Add 0.2ml of distilled water will yield a concentration of 500ug/ml. |
| Gene ID | 2010 |
|---|---|
| Other Names | Emerin, EMD, EDMD, STA |
| Calculated MW | 28994 MW KDa |
| Application Details | Immunohistochemistry(Paraffin-embedded Section), 0.5-1 µg/ml, Human, By Heat Western blot, 0.1-0.5 µg/ml, Human, Mouse, Rat |
| Subcellular Localization | Nucleus inner membrane ; Single-pass membrane protein; Nucleoplasmic side . Nucleus outer membrane. Colocalized with BANF1 at the central region of the assembling nuclear rim, near spindle-attachment sites. The accumulation of different intermediates of prelamin-A/C (non- farnesylated or carboxymethylated farnesylated prelamin-A/C) in fibroblasts modify its localization in the nucleus. |
| Tissue Specificity | Skeletal muscle, heart, colon, testis, ovary and pancreas. |
| Protein Name | Emerin |
| Contents | Each vial contains 5mg BSA, 0.9mg NaCl, 0.2mg Na2HPO4, 0.05mg NaN3. |
| Immunogen | A synthetic peptide corresponding to a sequence at the N-terminus of human Emerin (1-48aa MDNYADLSDTELTTLLRRYNIPHGPVVGSTRRLYEKKIFEYETQRRRL), different from the related mouse sequence by eight amino acids, and from the related rat sequence by nine amino ac |
| Purification | Immunogen affinity purified. |
| Cross Reactivity | No cross reactivity with other proteins |
| Storage | At -20˚C for one year. After r˚Constitution, at 4˚C for one month. It˚Can also be aliquotted and stored frozen at -20˚C for a longer time.Avoid repeated freezing and thawing. |
| Name | EMD |
|---|---|
| Synonyms | EDMD, STA |
| Function | Stabilizes and promotes the formation of a nuclear actin cortical network. Stimulates actin polymerization in vitro by binding and stabilizing the pointed end of growing filaments. Inhibits beta- catenin activity by preventing its accumulation in the nucleus. Acts by influencing the nuclear accumulation of beta-catenin through a CRM1- dependent export pathway. Links centrosomes to the nuclear envelope via a microtubule association. Required for proper localization of non- farnesylated prelamin-A/C. Together with NEMP1, contributes to nuclear envelope stiffness in germ cells (PubMed:32923640). EMD and BAF are cooperative cofactors of HIV-1 infection. Association of EMD with the viral DNA requires the presence of BAF and viral integrase. The association of viral DNA with chromatin requires the presence of BAF and EMD. |
| Cellular Location | Nucleus inner membrane; Single-pass membrane protein; Nucleoplasmic side. Nucleus outer membrane. Note=Colocalized with BANF1 at the central region of the assembling nuclear rim, near spindle-attachment sites. The accumulation of different intermediates of prelamin-A/C (non-farnesylated or carboxymethylated farnesylated prelamin-A/C) in fibroblasts modify its localization in the nucleus |
| Tissue Location | Skeletal muscle, heart, colon, testis, ovary and pancreas |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
Emerin is a serine-rich nuclear membrane protein that in humans is encoded by the EMD gene. And this gene is mapped to Xq28. Emerin is a member of the nuclear lamina-associated protein family. It mediates membrane anchorage to the cytoskeleton. Emery–Dreifuss muscular dystrophy is an X-linked inherited degenerative myopathy resulting from mutation in the EMD (also known clinically as STA) gene. Emerin appears to be involved in mechanotransduction, as emerin-deficient mouse fibroblasts failed to transduce normal mechanosensitive gene expression responses to strain stimuli. In cardiac muscle, emerin is also found complexed to beta-catenin at adherens junctions of intercalated discs, and cardiomyocytes from hearts lacking emerin showed beta-catenin redistribution as well as perturbed intercalated disc architecture and myocyte shape. This interaction appears to be regulated by glycogen synthase kinase 3 beta.
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.











 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.