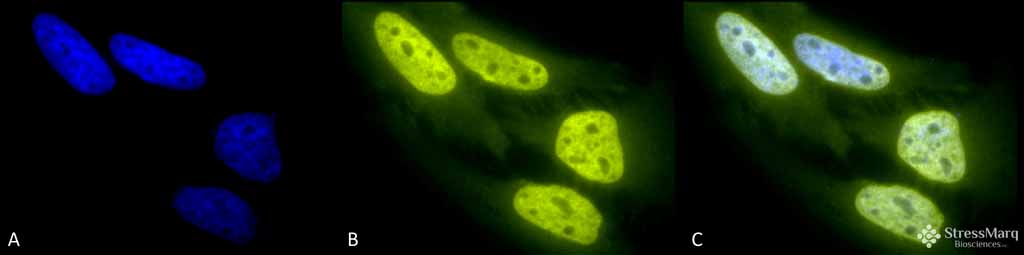
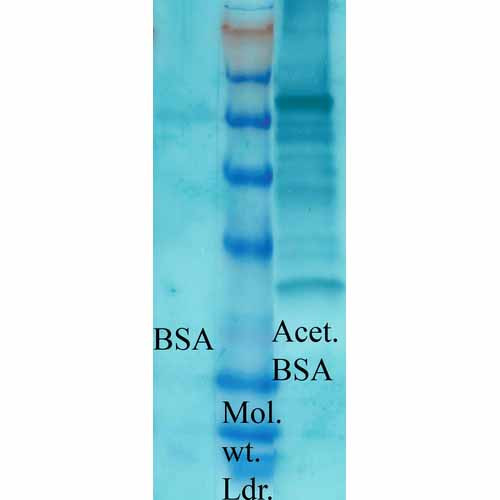
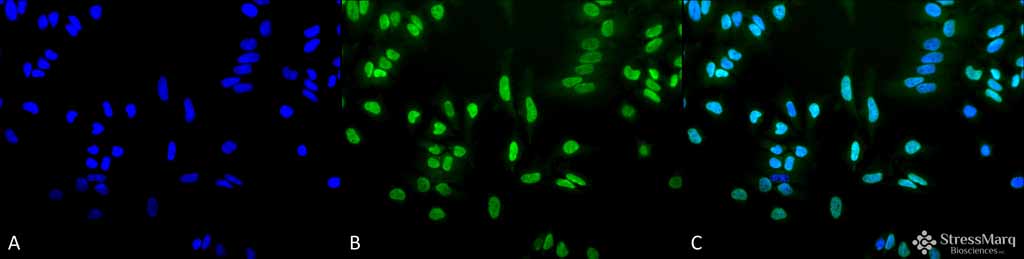
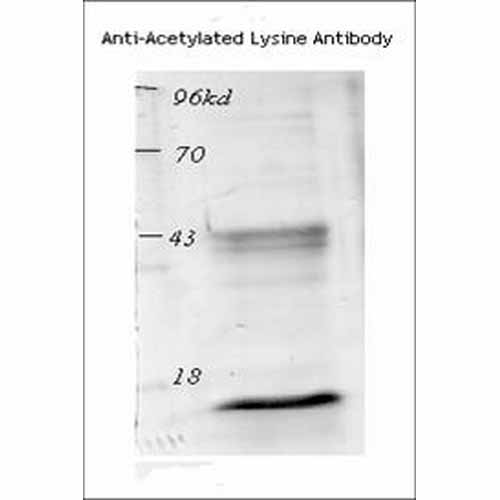
Acetylated Lysine Antibody
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB, IP, ICC |
|---|---|
| Host | Rabbit |
| Reactivity | Species Independent |
| Clonality | Polyclonal |
| Description | Rabbit Anti-Acetylated Lysine Polyclonal |
| Target/Specificity | Detects proteins containing acetylated lysine residues. No reaction to non-acetylated proteins. |
| Other Names | lysine Antibody, acetyl lysine Antibody |
| Immunogen | Acetylated KLH Conjugated |
| Purification | Protein A Purified |
| Storage | -20ºC |
| Storage Buffer | PBS, 50% glycerol, 0.09% sodium azide |
| Shipping Temperature | Blue Ice or 4ºC |
| Certificate of Analysis | A 1/250 dilution of SPC-155 was sufficient to detect the acetylated histone from TSA treated mouse spleen cell in western blot analysis. |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
Post-translational modifications of proteins play critical roles in the regulation and function of many known biological processes. Proteins can be post-translationally modified in many different ways, and a common post-transcriptional modification of Lysine involves acetylation (1). The conserved amino-terminal domains of the four core histones (H2A, H2B, H3 and H4) contain lysines that are acetylated by histone acetyltransferases (HATs) and deacetylated by histone deacetylases (HDACs) (2). Protein posttranslational reversible lysine Nε-acetylation and deacetylation have been recognized as an emerging intracellular signaling mechanism that plays critical roles in regulating gene transcription, cell-cycle progression, apoptosis, DNA repair, and cytoskeletal organization (3). The regulation of protein acetylation status is impaired in the pathologies of cancer and polyglutamine diseases (4), and HDACs have become promising targets for anti-cancer drugs currently in development (5).
References
1. Yang X.J. (2005) Oncogene. 24:1653-1662.
2. Hassig C.A. and Schreiber S.L. (1997) Curr. Opin. Chem. Biol. 1(3): 300-308.
3. Yang X.J. (2004) Bioessays 26:1076-1087.
4. Hughes R.E. (2002) Curr. Biol. 12: R141-R143.
5. Vigushin D.M. and Coombes R.C. (2004) Curr. Cancer Drug Targets 4: 205-218.
6. Chan H.M. et al. (2001) Nat. Cell Biol. 3: 667-674.
7. Martinez-Balbas M.A. et al. (2000) EMBO J. 19: 662-671.
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.




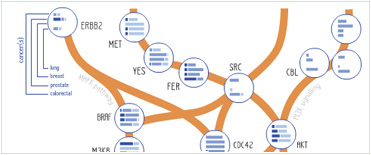







 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.