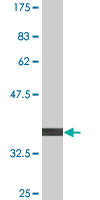
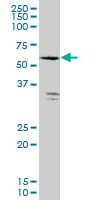
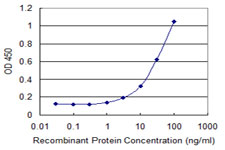
FLJ10986 Antibody (monoclonal) (M04)
Mouse monoclonal antibody raised against a partial recombinant FLJ10986.
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB, E |
|---|---|
| Primary Accession | Q96C11 |
| Other Accession | NM_018291 |
| Reactivity | Human |
| Host | mouse |
| Clonality | Monoclonal |
| Isotype | IgG2b Kappa |
| Clone Names | 3B9 |
| Calculated MW | 59993 Da |
| Gene ID | 55277 |
|---|---|
| Other Names | FGGY carbohydrate kinase domain-containing protein, 271-, FGGY |
| Target/Specificity | FLJ10986 (NP_060761, 1 a.a. ~ 99 a.a) partial recombinant protein with GST tag. MW of the GST tag alone is 26 KDa. |
| Dilution | WB~~1:500~1000 E~~N/A |
| Format | Clear, colorless solution in phosphate buffered saline, pH 7.2 . |
| Storage | Store at -20°C or lower. Aliquot to avoid repeated freezing and thawing. |
| Precautions | FLJ10986 Antibody (monoclonal) (M04) is for research use only and not for use in diagnostic or therapeutic procedures. |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
References
Personalized smoking cessation: interactions between nicotine dose, dependence and quit-success genotype score. Rose JE, et al. Mol Med, 2010 Jul-Aug. PMID 20379614.Analysis of DPP6 and FGGY as candidate genes for amyotrophic lateral sclerosis. Daoud H, et al. Amyotroph Lateral Scler, 2010 Aug. PMID 20001489.Analysis of FGGY as a risk factor for sporadic amyotrophic lateral sclerosis. Van Es MA, et al. Amyotroph Lateral Scler, 2009 Oct-Dec. PMID 19922138.Clinical and genetic features of patients with sporadic amyotrophic lateral sclerosis in south-west China. Fang DF, et al. Amyotroph Lateral Scler, 2009 Oct-Dec. PMID 19922123.Genomewide association study of movement-related adverse antipsychotic effects. Aberg K, et al. Biol Psychiatry, 2010 Feb 1. PMID 19875103.
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.











 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.