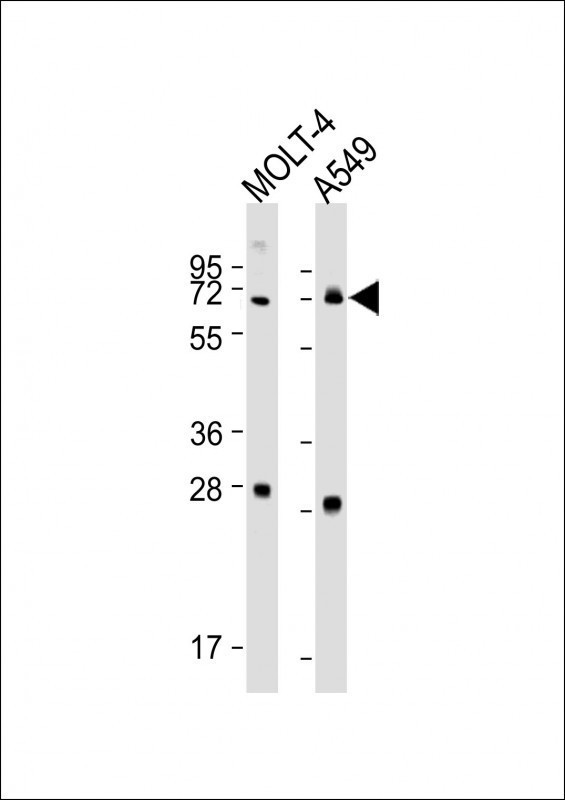
NEIL3 Antibody(Center)
Affinity Purified Rabbit Polyclonal Antibody (Pab)
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB, E |
|---|---|
| Primary Accession | Q8TAT5 |
| Other Accession | NP_060718.2 |
| Reactivity | Human |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | Rabbit IgG |
| Calculated MW | 67769 Da |
| Antigen Region | 292-320 aa |
| Gene ID | 55247 |
|---|---|
| Other Names | Endonuclease 8-like 3, 322-, DNA glycosylase FPG2, DNA glycosylase/AP lyase Neil3, Endonuclease VIII-like 3, Nei-like protein 3, NEIL3 |
| Target/Specificity | This NEIL3 antibody is generated from rabbits immunized with a KLH conjugated synthetic peptide between 292-320 amino acids from the Central region of human NEIL3. |
| Dilution | WB~~1:1000-1:2000 E~~Use at an assay dependent concentration. |
| Format | Purified polyclonal antibody supplied in PBS with 0.09% (W/V) sodium azide. This antibody is purified through a protein A column, followed by peptide affinity purification. |
| Storage | Maintain refrigerated at 2-8°C for up to 2 weeks. For long term storage store at -20°C in small aliquots to prevent freeze-thaw cycles. |
| Precautions | NEIL3 Antibody(Center) is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | NEIL3 |
|---|---|
| Function | DNA glycosylase which prefers single-stranded DNA (ssDNA), or partially ssDNA structures such as bubble and fork structures, to double-stranded DNA (dsDNA) (PubMed:12433996, PubMed:19170771, PubMed:22569481, PubMed:23755964). Mediates interstrand cross-link repair in response to replication stress: acts by mediating DNA glycosylase activity, cleaving one of the two N-glycosyl bonds comprising the interstrand cross-link, which avoids the formation of a double-strand break but generates an abasic site that is bypassed by translesion synthesis polymerases (By similarity). In vitro, displays strong glycosylase activity towards the hydantoin lesions spiroiminodihydantoin (Sp) and guanidinohydantoin (Gh) in both ssDNA and dsDNA; also recognizes FapyA, FapyG, 5-OHU, 5-OHC, 5-OHMH, Tg and 8-oxoA lesions in ssDNA (PubMed:12433996, PubMed:19170771, PubMed:22569481, PubMed:23755964). No activity on 8-oxoG detected (PubMed:12433996, PubMed:19170771, PubMed:22569481, PubMed:23755964). Also shows weak DNA-(apurinic or apyrimidinic site) lyase activity (PubMed:12433996, PubMed:19170771, PubMed:22569481, PubMed:23755964). In vivo, appears to be the primary enzyme involved in removing Sp and Gh from ssDNA in neonatal tissues (PubMed:12433996, PubMed:19170771, PubMed:22569481, PubMed:23755964). |
| Cellular Location | Nucleus. Chromosome {ECO:0000250|UniProtKB:A0A1L8HU22}. Note=Recruited to replication stress sites via interaction with ubiquitinated CMG helicase {ECO:0000250|UniProtKB:A0A1L8HU22} |
| Tissue Location | Expressed in keratinocytes and embryonic fibroblasts (at protein level). Also detected in thymus, testis and fetal lung primary fibroblasts. |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
NEIL3 belongs to a class of DNA glycosylases homologous to the bacterial Fpg/Nei family. These glycosylases initiate the first step in base excision repair by cleaving bases damaged by reactive oxygen species and introducing a DNA strand break via the associated lyase reaction (Bandaru et al., 2002 [PubMed 12509226]).
References
Krokeide, S.Z., et al. Protein Expr. Purif. 65(2):160-164(2009)
Takao, M., et al. Genes Cells 14(2):261-270(2009)
Dallosso, A.R., et al. Gut 57(9):1252-1255(2008)
Bethke, L., et al. J. Natl. Cancer Inst. 100(4):270-276(2008)
Newton-Cheh, C., et al. BMC Med. Genet. 8 SUPPL 1, S7 (2007) :
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.











 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.