HSP22 Antibody
HSP22 Antibody, Clone 3C12-H11
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB, IHC, ICC, E |
|---|---|
| Primary Accession | Q9UJY1 |
| Other Accession | NP_055180.1 |
| Host | Mouse |
| Isotype | IgG1 Kappa |
| Reactivity | Human, Mouse, Rat |
| Clonality | Monoclonal |
| Description | Mouse Anti-Human HSP22 Monoclonal IgG1 Kappa |
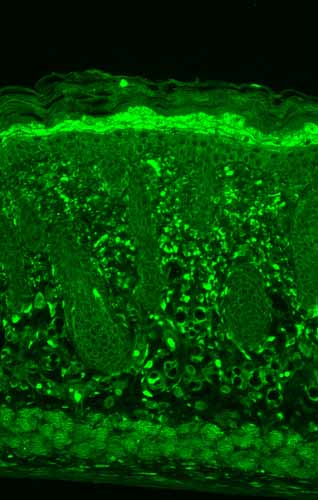
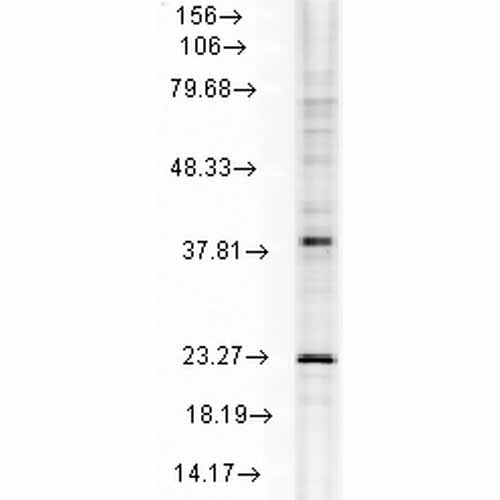
| Target/Specificity | Detects ~22kDa. Detects endogenous and exogenous HSP22 in monomeric, dimeric and tetrameric forms in WB. Does not cross react with alpha crystallin. |
| Other Names | Alpha crystallin C chain Antibody, CMT2L Antibody, CRYAC Antibody, DHMN2 Antibody, H11 Antibody, Heat shock 22kDa protein 8 Antibody, HMN2 Antibody, HSB8 Antibody, HSPB8 Antibody |
| Clone Names | 3C12-H11 |
| Immunogen | His-tagged human recombinant HSP22 |
| Purification | Protein G Purified |
| Storage | -20ºC |
| Storage Buffer | PBS pH7.4, 50% glycerol, 0.09% sodium azide |
| Shipping Temperature | Blue Ice or 4ºC |
| Certificate of Analysis | 1 µg/ml of SMC-187 was sufficient for detection of HSP22 in 20 µg of whole rat tissue extract by ECL immunoblot analysis using Goat anti-mouse IgG:HRP as the secondary antibody. |
| Cellular Localization | Cytoplasm | Nucleus |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
HSP27s belong to an abundant and ubiquitous family of small heat shock proteins (sHSP). It is an important HSP found in both normal human cells and cancer cells. The basic structure of most sHSPs is a homologous and highly conserved amino acid sequence, with an α-crystallin domain at the C-terminus and the WD/EPF domain at the less conserved N-terminus. This N-terminus is essential for the development of high molecular oligomers (1, 2). HSP27-oligomers consist of stable dimers formed by as many as 8-40 HSP27 protein monomers (3). The oligomerization status is connected with the chaperone activity: aggregates of large oligomers have high chaperone activity, whereas dimers have no chaperone activity (4). HSP27 is localized to the cytoplasm of unstressed cells but can redistribute to the nucleus in response to stress, where it may function to stabilize DNA and/or the nuclear membrane. Other functions include chaperone activity (as mentioned above), thermo tolerance in vivo, inhibition of apoptosis, and signal transduction. Specifically, in vitro, it acts as an ATP independent chaperone by inhibiting protein aggregation and by stabilizing partially denatured proteins, which ensures refolding of the HSP70 complex. HSP27 is also involved in the apoptotic signaling pathway because it interferes with the activation of cytochrome c/Apaf-1/dATP complex, thereby inhibiting the activation of procaspase-9. It is also hypothesized that HSP27 may serve some role in cross-bridge formation between actin and myosin (5). And finally, HSP27 is also thought to be involved in the process of cell differentiation. The up-regulation of HSP27 correlates with the rate of phosphorylation and with an increase of large oligomers. It is possible that HSP27 may play a crucial role in termination of growth (6).
References
1.Kappe G., et al. (2001) Biochem Biophys Acta 1520: 1-6.
2. Benndorf R., et al. (2001) J Biol Chem 276: 26753-26761.
3.Sun X., et al. (2004) J Biol Chem 279: 2394-2402.
4.Kim M.V., et al. (2004) Biochem Biophys Res Commun 325: 649-652.
5. Wilhelmus M.M., et al. (2006)Acta Neuropathol (Berl) 111: 139-149.
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.




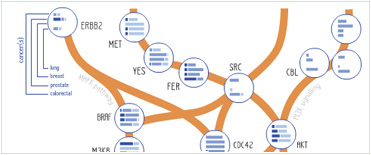







 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.